Medical devices
Alzheimer's device startup raises $105 million
%20(1).png?width=1252&height=834&upscale=true&name=Spectris%20device%202%20(2)%20(1).png)
Cognito Therapeutics, which is working on a light and sound stimulus device to treat Alzheimer's disease, raised $105 million lead by Morningside Ventures and a cadre of other investors. The company is awaiting the results of its pivotal trial which will read out later this year. It also expects to file to the Food and Drug Administration for de novo marketing authorization by the end of the year.
Cognito's product, called Spectris, looks a bit like sunglasses attached to a pair of AirPods Max and could prove an attractive (and stylish???) alternative to the approved drug therapies for Alzheimer's that come with potentially dangerous side effects. But as I report in my story, the treatment that's meant to be used at home in perpetuity may require new reimbursement and care pathways.
Read more here
regulation
FDA notes: 30 warnings and an AI approval
- FDA on Tuesday sent 30 warning letters to telehealth companies, including Good GirlRx, Skinny Rx, and WeightCare, for making false or misleading claims regarding compounded GLP-1 products offered on their websites. This follows 50 letters sent in the fall.
- FDA this week approved Claire, an AI-enabled device from Perimeter Medical Imaging that identifies and marks areas suspicious for breast cancer during surgery to help ensure the procedure removes all diseased tissue. Unlike most of the AI-enabled devices we talk about around here, Claire was authorized through the premarket approval pathway reserved for the highest-risk devices.
medical devices
Another notable breakthrough device
Radiology foundation model developer
Cognita (not to be confused with Cognito above),
announced it received a breakthrough device designation from FDA for Cognita Chest X-Ray, its vision-language model that interprets images
and drafts radiology reports for clinician review. The latter is an ambitious milestone on the roadmap for radiology AI companies, and it's among the generative AI applications that will pose challenges for FDA. Cognita was recently acquired for $80 million by
Mosaic Clinical Technologies, a subsidiary of
Radiology Partners, which operates over 3,000 radiology sites.
Katie Palmer wrote about Cognita in the fall.
This is the second generative AI breakthrough device
we've heard about this week. We don't make a habit of covering every breakthrough device announcement because FDA issues tons of them — 149 in 2025 alone. But it's interesting to me that we're starting to hear about a concrete pipeline of generative AI-based medical devices heading into the FDA. If you've got those, send them our way.
DIgital health
Digital GI solutions get the thumbs up
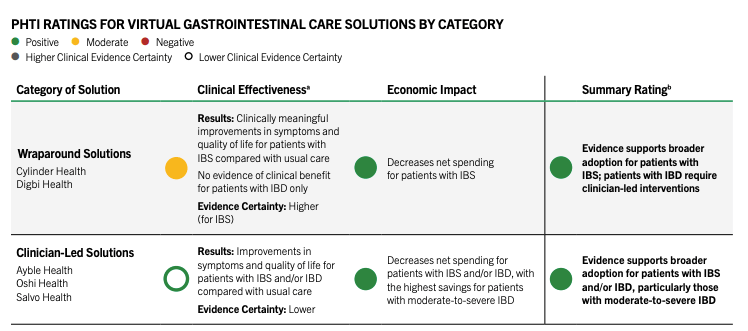
The Peterson Health Technology Institute's latest assessment looks at the clinical effectiveness and economic impact of digital gastrointestinal care solutions from startups including Ayble Health, Oshi Health, Salvo Health, and more. PHTI gave the solutions a resounding endorsement after estimating two different care models can save over $1,000 per user by helping to avoid hospitalizations and other high-cost services.


No comments