medical devices
CMS may nix breakthrough device payment pathway
Medicare regulators are proposing to repeal a payment pathway available to medical devices used by hospitals if they are designated as 'breakthrough' by the Food and Drug Administration. If finalized, the policy would require breakthrough devices, like other new technologies, to show substantial improvement over the status quo to qualify for those supplementary payments. Currently, the products must only show that they are expensive.
The FDA breakthrough device pathway helps speed up the regulatory process for experimental products that are novel and fill unmet needs. However, companies complain that it's still too difficult to get devices paid for once they are on the market. The first Trump administration granted the current payment pathway, and industry hoped the new administration would push things further. Medicare appears to be going in the opposite direction. “As we have gained experience, we have concerns with the limited evaluation process" for the supplemental payments, regulators wrote in the proposed rule. I expect a lot of pushback.
Read the full story from Katie Palmer here
artificial intelligence
Are CT calcium screenings worth it?
Katie has a very interesting story about the rise of opportunistic coronary artery calcium screening, which uses AI to examine chest CT scans taken for other reasons, and can help identify people who may benefit from statins. Or I should say, the potential rise in the use of these algorithms. The technology exists, is marketed by several companies, and Medicare just set a payment rate for it. But so far the uptake has been modest as hospitals weigh the benefits and risks.
The upside: Hospitals can now get paid $15 a pop for some screenings and they may help some patients. The downside is there’s no definitive evidence screenings lead to better long-term health outcomes. On the flip side, the screenings could kick off more risky follow-up testing, and specialist consultations at a time when cardiologists are already overloaded with patients. Applying the new tech without careful guidelines could result in increased demand for clinicians that are in scarce supply.
Read more here
telehealth
Katie writes: The Department of Justice filed an amended complaint against Cerebral co-founder Kyle Robertson last week, alleging patterns of deceptive billing at his newer telehealth company, Zealthy. Cerebral fell into the DOJ’s crosshairs for prescribing stimulants like Adderall en masse; Zealthy, like so many other telehealth companies in the last 4 years, has built its business on GLP-1s.
The complaint also alleges that Zealthy used the name and provider identifier of a doctor — a former medical director not involved in medical care at the company — to order thousands of prescriptions without his knowledge. The government requested an asset freeze and receivership for Zealthy “to halt Robertson and Zealthy’s runaway campaign of lawbreaking, which is actively deceiving telehealth patients, endangering their safety, and raiding their bank accounts.”
Read STAT's coverage of Robertson's previous troubles here.
data
Despite telemental health boom, in-person care rules
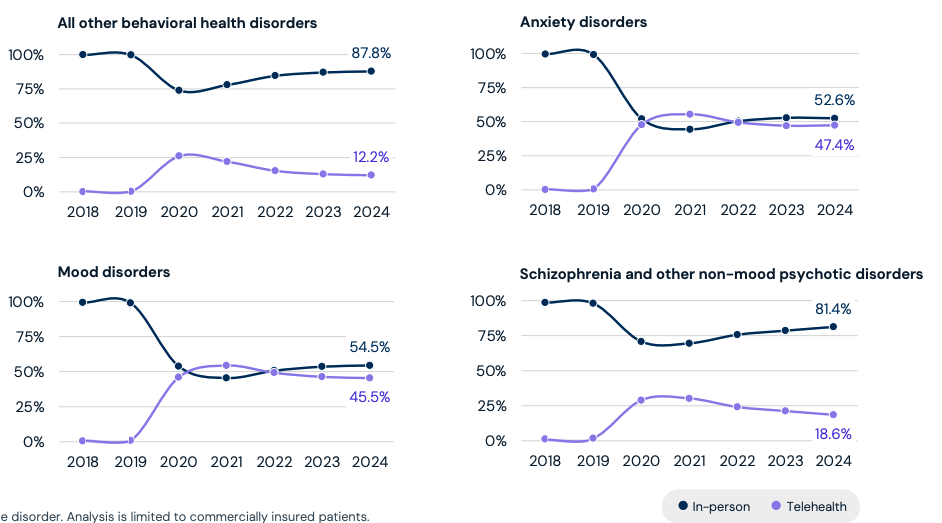
By now we know that behavioral health was the big winner of the telehealth boom during the pandemic. It accounted for 66% of telehealth claims to commercial payers in 2024 according to a new report from health care analytics and intelligence company Trilliant Health.
While in-person care is now most common for all behavioral health specialties, nearly half of anxiety treatment happens over telehealth, which in turn accounts for 58% of all behavioral health telehealth use.


No comments